L’oro come “bene di rifugio”
L’oro costituisce a volte parte di un investimento finanziario difensivo (bene rifugio per la tutela del capitale), data la stabilità del suo valore commerciale a lungo termine e la sua sostanziale scorrelazione rispetto all’andamento del mercato azionario ed obbligazionario; proprio per questa sua stabilità, la speculazione sull’oro diventa particolarmente appetibile quando la fiducia in una valuta viene meno, e quando il valore di una valuta è soggetto ad iperinflazione.
Il prezzo dell’oro è anche alla base di futures con cui si specula sul suo ipotizzato valore futuro. Il valore dell’oro è fortemente influenzato dall’offerta, motivo per cui la sua estrazione è ponderata attentamente: incrementarne la produzione significa spesso farne crollare il prezzo.


Carature dell'oro
Nelle leghe d’Oro il termine carato viene indicato come unità di misura proporzionale di alla “purezza” che quantifica le parti d’oro in una lega su base 24/24. Il “carato” nelle leghe d’oro equivale ad una parte d’oro su un totale di 24 parti di metallo costituente la lega. Ne deriva, ad esempio, che la dicitura 18 carati sta ad indicare che la lega è costituita da 18 parti d’oro fino e 6 parti di altri metalli e viene abbreviato con le sigle ct o kt o prevalentemente con la sola k spesso affiancata al numero senza alcun spazio intermedio, ad esempio 18k.
L’oro di massima purezza è dunque a 24 carati (24 parti d’oro “fino” su 24 totali) e si indica con la sigla 24k.
- 9 ct ( 333 parti di oro su 1000 )
- 12 ct ( 500 parti di oro su 1000 )
- 14 ct ( 585 parti di oro su 1000 )
Dai 9 carati ai 14 carati sono utilizzati da diverse Nazioni per la produzione di gioielli, ad esempio provenienti dall’America o dalla Germania.
- 18 ct ( 750 parti di oro su 1000 )
I 18 carati sono i più comuni usati in Italia dalle fabbriche e dagli artigiani Orafi per produrre gioielli.
- 22 ct ( 916 parti di oro su 1000 )
I 22 carati vengono utilizzati per le Monete tipo Sterlina, Marenghi e altre.
- 24 ct ( 999 parti di oro su 1000 )
I 24 carati vengono utilizzati per la produzione di Lingotti e alcune Monete.Accanto, nella maggior parte dei casi, c’è un ulteriore timbro che rappresenta l’ “autografo” identificativo dell’orafo che ha creato l’oggetto. Questo timbro rappresenta l’identificazione del laboratorio nel quale è stato creato l’oggetto. ( Es. tipo “1-AR”, ovvero, Arezzo – fabbrica numero uno o, “45-VI” (laboratorio n. 45 di Vicenza) ). Molti paesi esigono che i gioielli in vero oro abbiano un marchio di garanzia. Questo significa che quando l´oggetto è prodotto, riceve un timbro ufficiale che indica il grado di oro che è stato usato nell’oggetto. Di solito il timbro è posto in un punto non appariscente come l´interno di un anello o sul retro della chiusura di un bracciale.
Leghe d’oro
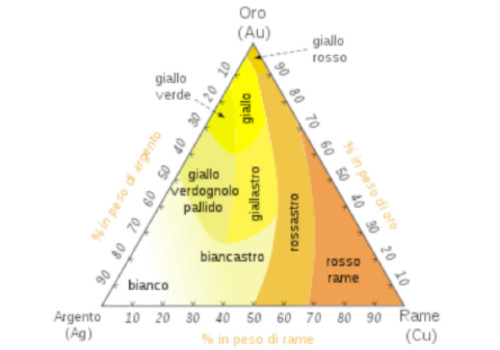
L’oro da gioielleria, cioè quello legato a uno o più metalli per aumentarne la rigidità, presenta una colorazione bianca o rossa, a seconda del tipo di lega (con argento o rame).
L’oro giallo è composto al 75% d’oro, al 12,5% d’argento e al 12,5% da rame.
L’oro rosa è normalmente composto dal 75% d’oro, al 5% d’argento ed al 20% da rame.
L’oro grigio è composto al 75% d’oro, al 15% d’argento ed al 10% di rame.
L’oro blu è una lega di oro e di ferro. Un trattamento termico ossida gli atomi di ferro sulla superficie dell’oro, e gli dona la colorazione azzurra.
L’oro bianco da gioielleria è composto al 75% da oro, ed al 25% da nichel, argento o palladio.
Bisogna notare che il termine “oro bianco” è spesso utilizzato per designare l’oro grigio in bigiotteria. L’oro bianco è ricoperto da un fine strato di rodio, che sparisce per usura, con il tempo, ridando un colore giallo all’oro. È una lega inventata dopo la prima guerra mondiale. Per la doratura tramite fogli sottili di oro, la lega deve essere il più possibile duttile e malleabile.
L’oro giallo da doratura è composto dal 98,0% d’oro, dall’1,0% d’argento e dall’1,0% da rame. Può anche essere puro.
L’oro rosso da doratura è composto dal 94,5% d’oro e dal 5,5% di rame.
L’oro ½ giallo da doratura è composto dal 91,5% d’oro, dal 6,0% d’argento e dal 2,5% di rame.
L’oro limone da doratura è composto dal 94,5% d’oro e dal 5,5% d’argento.
L’oro grigio da doratura è composto dal 75,5% d’oro, 14,5% di palladio e dal 10,0% d’argento.
L’oro bianco francese da doratura è composto dal 20,0% d’oro e dall’80,0% d’argento, altrove in Europa, è al 50,0% oro, e al 50,0% argento.
L’oro ed il suo utilizzo nei diversi settori
L’oro puro è troppo tenero per poter essere lavorato normalmente; viene indurito legandolo ad altri metalli (in genere rame e argento). L’oro e le sue leghe sono usati in gioielleria, nel coniare monete e sono uno standard di cambio valutario per molte nazioni. Per via della sua resistenza alla corrosione ed alle sue notevoli proprietà elettriche, ha trovato sempre più spazio anche in applicazioni industriali. Sono in corso studi sull’utilizzo dell’oro come catalizzatore; infatti l’oro mostra una grande attività catalitica quando si trova in forma di nanoparticelle disperse su adeguati supporti.
I catalizzatori supportati a base di oro ricoprono un ruolo fondamentale in diverse reazioni, tra cui:
ossidazione di monossido di carbonio (CO);
ossidazione completa di idrocarburi;
ossidazioni selettive;
reazioni di water-gas shift.
L’attività di questi catalizzatori dipende notevolmente dal metodo di preparazione e dal tipo di supporto utilizzato.
Tra gli altri usi:
- nella componentistica elettronica:
svolge funzioni critiche in molti computer, apparecchi per telecomunicazioni, motori jet e numerose applicazioni industriali;
trova ampio uso come materiale di rivestimento delle superfici di contatti elettrici, per garantirne la resistenza alla corrosione nel tempo;
- in ambito astronautico:
l’oro è usato come rivestimento protettivo di molti satelliti artificiali, data la sua elevata capacità di riflettere sia la luce visibile che quella infrarossa;
- a scopo medico e diagnostico:
l’oro colloidale viene utilizzato in un particolare tipo di elettroforesi, una metodica di diagnostica medica.
realizzazione di otturazioni e ponti in odontoiatria;
in sospensione colloidale, trova impiego nella pittura delle ceramiche ed è oggetto di studio per applicazioni biologiche e mediche;
l’aurotiomaleato di disodio è un farmaco per la cura dell’artrite reumatoide;
l’isotopo radioattivo 198Au (emivita: 2,7 giorni) è usato in alcune terapie anti-tumorali;
- nelle indagini a miscroscopio:
l’oro è usato per rivestire campioni biologici da osservare sotto un microscopio elettronico a scansione;
- in ambito fotografico:
l’acido cloroaurico trova uso in fotografia per far virare l’immagine prodotta dai sali d’argento;
- a scopo ornamentale:
l’oro può essere tirato in fili ed inserito in tessuti ed ornamenti;
l’oro bianco – una lega con platino, palladio, nichel o zinco – funge da sostituivo del platino in alcune applicazioni ed in gioielleria; – inoltre tale lega ha un basso coefficiente di dilatazione termica;
l’oro verde (in lega con l’argento) e l’oro rosso (in lega col rame) sono usati in gioielleria;
- nello sport e nelle premiazioni:
una medaglia d’oro è il premio assegnato al vincitore di moltissime competizioni e concorsi, non ultimi le Olimpiadi ed il Premio Nobel;
- nella cucina:
l’oro metallico è usato come ingrediente in alcune ricette di alta cucina; non avendo praticamente alcuna reattività non altera i sapori.



